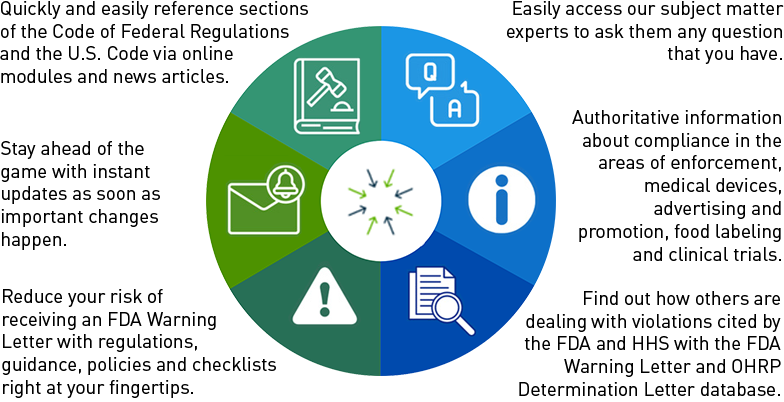
Joint FDA-OHRP Draft Guidance Details ‘Key Information’ in Informed Consent
March 11, 2024 at 11:42 AM EST
The FDA and the Department of Health and Human Services’ Office for Human Research Protections (OHRP) released joint draft guidance March 1 addressing the revised Common Rule requirement to begin informed consent with “key information” to aid subject understanding.
The draft guidance provides recommendations for developing a key information section for clinical trials or stud... Read More